Modification Of Zn-Cr Layered Double Hydroxide With Keggin Ion [?-SiW12O40]4- AS Cr(VI) Adsorbent
Abstract
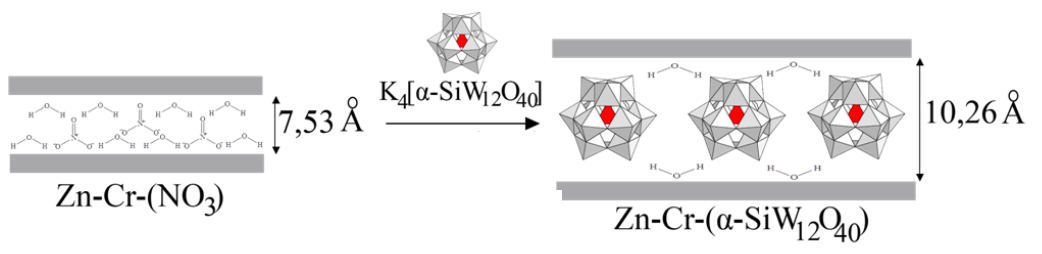
Abstract: In this study, synthesis of layered double hydroxide of Zn-Cr and Zn-Cr intercalated with keggin ion [SiW12O40]4- have been carried out. The synthesized material was characterized using XRD, BET, and FTIR. Material of Zn-Cr layered double hydroxide and the intercalation results are used as Cr(VI) ion adsorbent. Factors that influence the adsorption process studied to obtain kinetic and thermodynamic parameters include time, concentration and temperature variation of adsorption. Based on XRD characterization, the distance between layers from 7,53 ? on the Zn-Cr layered double hydroxide before intercalation to 10,26 ? on Zn-Cr layered double hydroxide intercalated with keggin ion [SiW12O40]4-. BET analysis showed that the surface area of both materials increased from 31,638 m2/g to 128,871 m2/g. The result of pH Point Zero Charge measurement for the Zn-Cr layered double hydroxide material is 10 while the Zn-Cr intercalated with keggin ion [SiW12O40]4- material is 8. Based on the kinetics model, adsorption of Cr(VI) ion follows pseudo-second-order model with a linear regression coefficient close to 1. Furthermore, the intercalation of the Zn-Cr intercalated with keggin ion [SiW12O40]4- shows the higher adsorption capacity for the Cr(VI) ion than the Zn-Cr layered double hydroxide as a control.
Keywords: adsorption, Cr(VI), keggin ion, layered double hydroxide, Zn-Cr